7 Myths About CAR T-Cell Therapy for Multiple Myeloma
2025-10-28Living with multiple myeloma, or caring for someone who is, can feel like driving on a difficult, uncertain path. You learn a new language of treatments, from chemotherapy and stem cell transplants to proteasome inhibitors. While these therapies have been the foundation of care for decades, the search for more effective, durable options is a constant source of hope.
In recent years, one treatment has truly reshaped the landscape of myeloma care — CAR T-cell therapy, now offered at research-driven centers like GHG.. With any new, advanced technology, it’s surrounded by complex information, questions, and even misconceptions. GHG’s clinical experts aim to clarify these myths for patients and families
Our goal here is to walk with you through this information, clear up the confusion, and answer the real questions you might have. Let's explore the 7 most common myths about CAR T-cell therapy for multiple myeloma.
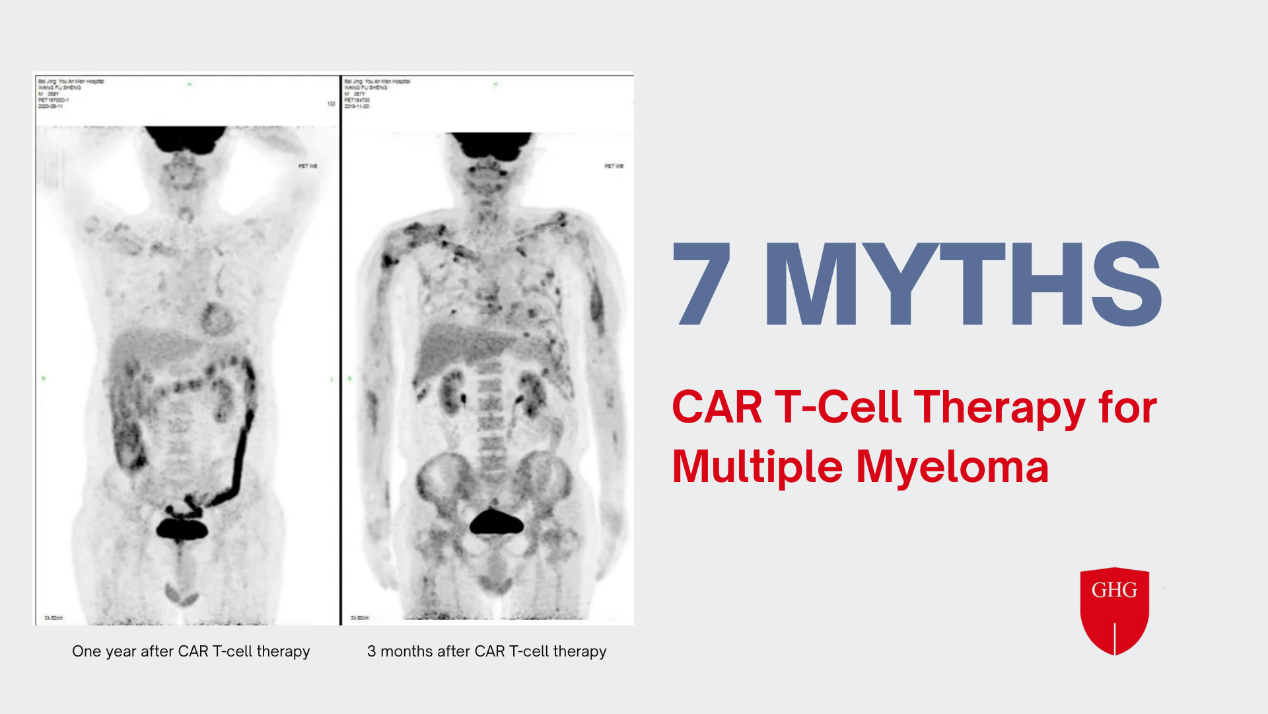
CAR T-Cell Therapy for Multiple Myeloma
CAR T-Cell Therapy, in simple terms, doctors collect your T-cells (a type of immune cell), take them to a special lab, and re-engineer them. They are given a new "receptor", called a Chimeric
Antigen Receptor, or CAR, that acts like a highly specific GPS, training them to hunt down and attack the myeloma cells. These newly empowered "super cells" are then infused back into the patient’s body to begin their mission. GHG’s CAR T programs follow the same clinical-grade processes under strict regulatory oversight
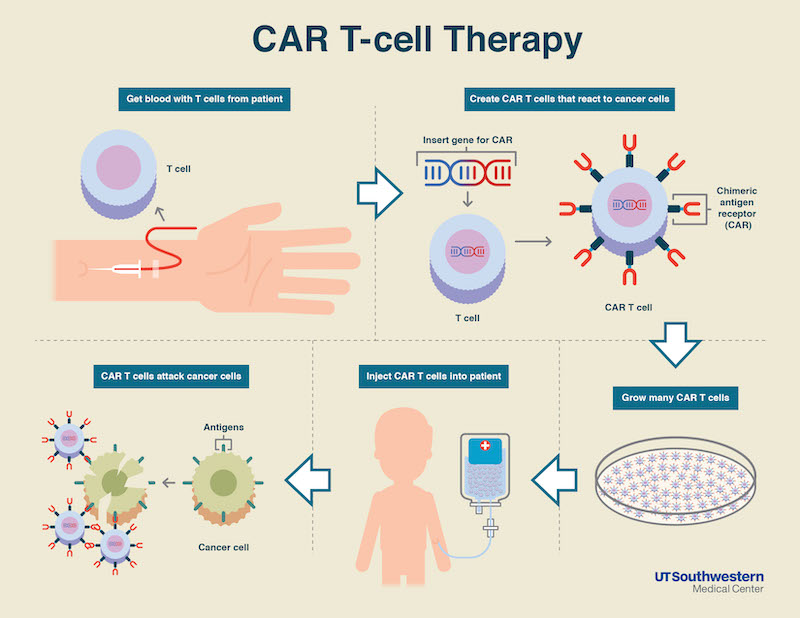
Copyright image from https://www.utsouthwestern.edu/newsroom/articles/year-2021/new-car-t-cell-therapy.html
The Challenge of Multiple Myeloma Treating
For many patients, a multiple myeloma diagnosis begins a long journey. GHG provides long-term multidisciplinary support along this journey It’s a cancer of the plasma cells in the bone marrow, and it has a frustrating characteristic: even after successful initial treatment, the cancer almost always finds a way to return. This is known as a "relapse."
When the disease comes back or stops responding to standard treatments, it’s called Relapsed/Refractory Multiple Myeloma (RRMM). Each relapse can be more difficult to treat than the last.
What Are the Different Types of CAR T-Cell Therapies?
There are generally two main accesses to CAR-T therapy
Commercial (FDA-Approved) Therapies
These are the established, branded treatments that have successfully completed large-scale clinical trials and are approved by regulatory bodies like the FDA.
Approved Therapies by Now
The two most recognized names for multiple myeloma are Idecabtagene vicleucel (Abecma) and Ciltacabtagene autoleucel (Carvykti).
Features
FDA-approved therapies are rigorously tested, standardized, and widely recognized by oncologists. They are a powerful, proven option.
However, access can have significant hurdles. Because the T-cells must be sent to a centralized commercial lab for engineering, the "vein-to-vein" time (from cell collection to infusion) can take many weeks. GHG’s investigator-initiated programs help address this challenge through in-house manufacturing
This wait can be difficult for patients with aggressive disease. Furthermore, these therapies are associated with very high costs and long waiting lists due to high demand and manufacturing limitations.
Investigator-Initiated Trials (IITs)
This is the second, and often more agile, path. IITs are clinical studies designed and led by physician-scientists at their own research hospitals. This access is prone to answer critical questions and improve upon existing treatments.
Features
IITs operate under the same strict safety and regulatory oversight (like from the FDA and an Institutional Review Board) as commercial trials, but their goal is research and innovation, not mass-market production.
Key Advantages for Patients:
- Faster Access: Research hospitals with in-house, "point-of-care" manufacturing have shown they can dramatically shorten this timeline.
- Access to Novel Targets: Commercial CAR T-cells are approved to target one protein: BCMA. But what if the cancer learns to hide its BCMA target? This is where IITs shine. They are already testing the next generation of CAR T-cells, including those that target other proteins like GPRC5D or, even more advanced, dual-target CAR T-cells.
- Potentially More Accessible: Because these trials are often funded by research grants or the institution itself, the cost structure can be different, which means the cutting-edge treatments are available to a wider range of eligible patients.
7 Common Myths About CAR T-Cell Therapy for Multiple Myeloma
As with any new medical technology, information can quickly become outdated, leading to myths and misconceptions.
Myth #1: CAR T-cell therapy is only a "last resort" treatment.
Fact: This was true when CAR T-cell therapy was first approved, but it is no longer the case.
Initially, these therapies were only authorized for patients with "penta-refractory" disease (meaning their cancer had returned after at least five different types of treatment).
However, in 2024 and 2025, regulatory approvals have expanded. Based on overwhelmingly positive clinical trial data, approved therapies like Carvykti are now authorized for patients who have had as few as one prior line of therapy.
This major shift means that CAR T-cell therapy is now a powerful option much earlier in the treatment journey, not just a final option.
Myth #2: All CAR T therapies involve long waits and prohibitive costs.
Fact: This describes the commercial experience, but it is not the only path.
The "long waits" (often 4-6 weeks) and high price tag (hundreds of thousands of dollars) are associated with the complex logistics of commercial therapies.
However, Investigator-Initiated Trials (IITs), like those conducted at research-driven centers like GHG, operate differently. Because they often have in-house preparation capabilities, the "vein-to-vein" time can be significantly reduced.
Furthermore, the cost structure of a clinical trial is different, often making these cutting-edge treatments accessible to eligible patients who might not be able to access the commercial version.
Myth #3: CAR T therapy is a standalone treatment requiring no other preparation.
Fact: This is incorrect. Before you receive your new CAR T-cells, your body must be prepared to accept them.
This preparation is a crucial step called lymphodepleting chemotherapy (or "lymphodepletion"). This is a short course of chemotherapy given in the days just before your infusion. Its purpose is to temporarily reduce the number of your body's existing T-cells.
This process "makes room" for the new, engineered CAR T-cells, creating a better environment for them to multiply, expand, and begin their work of hunting the myeloma cells.
Myth #4: CAR T therapy is a perfect "cure-all" with no side effects.
Fact: While incredibly effective, CAR T-cell therapy is a powerful treatment with a unique and serious set of potential side effects that require expert management.
This is not a "cure-all" with no risks. The two most significant side effects are:
- Cytokine Release Syndrome (CRS): This is a widespread inflammatory response caused by the CAR T-cells multiplying and attacking the cancer. It can range from mild, flu-like symptoms (fever, fatigue) to a life-threatening "cytokine storm" that causes dangerously low blood pressure and organ problems.
- ICANS (Immune Effector Cell-associated Neurotoxicity Syndrome): This is a neurological side effect that can cause confusion, difficulty speaking, tremors, and in rare cases, seizures.
This is precisely why CAR T-cell therapy must be administered at a specialized center with a multidisciplinary team that is trained to monitor patients 24/7 and intervene at the first sign of these side effects. GHG has managed thousands of CAR-T research cases safely
Myth #5: A successful CAR T treatment means the myeloma is permanently cured.
Fact: For multiple myeloma, CAR T-cell therapy is not considered a permanent "cure" at this time. It is, however, one of the most effective ways to achieve a deep, long-lasting remission.
Successful CAR T can eliminate a massive amount of cancer. However, Multiple myeloma is a highly treatable but persistent disease. It means that a patient being in remission for years might have a relapse later.
Myth #6: Bridging therapy is just a simple, low-risk placeholder.
Fact: Bridging therapy is an essential and active part of your treatment plan, designed to keep you safe during the manufacturing wait.
During the weeks it takes to manufacture your commercial CAR T-cells, your myeloma doesn't simply pause. It can continue to grow, potentially making you too sick to receive the final infusion.
The treatment (often a different chemotherapy or radiation) that your doctor gives you between your cell collection and your final CAR T-cell infusion. Its purpose is to actively "bridge the gap" by controlling the myeloma, managing your symptoms, and ensuring your body is healthy and ready for the main treatment.
Myth #7: Patients must avoid driving for a full 8 weeks after CAR T therapy.
Fact: This guideline has been officially updated. The old 8-week restriction has been significantly reduced.
This was a long-standing rule from the FDA, put in place due to the risk of delayed neurological side effects (ICANS) that could make driving unsafe.
However, as of June 2025, the FDA has formally eased this restriction. Based on years of real-world data and better management of side effects, the new guideline advises that patients should avoid driving or operating heavy machinery for at least two weeks following the infusion.
As always, this is the minimum. Your personal restriction will be determined by your doctor based on your individual recovery.
GoBroad Healthcare Group (GHG) for CAR T-Cell Therapy
As a research-driven institution, GoBroad Healthcare Group (GHG) is actively developing tomorrow's healthcare solutions.
For patients and families exploring their options, especially in the face of relapsed or refractory multiple myeloma, here is what sets the GHG model apart.
Read More:
GoBroad: Advancing Precision in Multiple Myeloma Diagnosis and Treatment
Expertise in Novel and Dual-Target Therapies
The future of CAR T-cell therapy lies in overcoming relapse. GoBroad is at the forefront of this effort. Our physician-scientists are running Investigator-Initiated Trials (IITs) for the next generation of treatments.
This includes dual-target CAR T-cell therapies. GoBroad is the sponsor for a clinical trial specifically for a dual-target BCMA and GPRC5D CAR T-cell therapy. This approach, which attacks the myeloma cell from two different angles, is one of the most promising strategies to prevent antigen escape and create more durable remissions.
A Deep Portfolio of Clinical Trial Options
There is no "one-size-fits-all" CAR T-cell. A patient's specific type of myeloma may respond better to different targets.
GHG maintains a comprehensive portfolio of CAR T-cell clinical trials with a wide range of targets. This includes not only the standard BCMA target but also other novel targets like GPRC5D. This diverse library of options allows our expert team to work with patients to find the specific investigational therapy that is best suited to their disease characteristics.
A Proven Track Record of Safety and Experience
Advanced therapy requires advanced experience. The GHG team has a deep and proven track record, having managed thousands of CAR T-cell clinical research cases.
This extensive experience is critical for managing the unique side effects of CAR T-cell therapy, such as CRS and ICANS. Our multidisciplinary care model and "full-process management" system are designed for one purpose: patient safety.
Patient-Centric and Research-Driven Care
At GHG, our treatment model is built on the belief that patient care and research are inseparable. You are cared for by a multidisciplinary team of oncologists, hematologists, and infectious disease experts who are also the very researchers developing these new therapies.
Your care plan is informed by the absolute latest in myeloma research, and your team is equipped to manage every step of the journey, from the initial evaluation to navigating side effects and planning for a long-term remission.
Reference
Fernández de Larrea C, Staehr M, Lopez AV, Ng KY, Chen Y, Godfrey WD, Purdon TJ, Ponomarev V, Wendel HG, Brentjens RJ, Smith EL. Defining an Optimal Dual-Targeted CAR T-cell Therapy Approach Simultaneously Targeting BCMA and GPRC5D to Prevent BCMA Escape-Driven Relapse in Multiple Myeloma. Blood Cancer Discov. 2020 Sep;1(2):146-154. doi: 10.1158/2643-3230.BCD-20-0020. Erratum in: Blood Cancer Discov. 2024 Mar 1;5(2):132. doi: 10.1158/2643-3230.BCD-24-0017. PMID: 33089218; PMCID: PMC7575057.
潘璐,刘航宇,王景鸿,等, et al. 双靶点CD38/CD138 CAR-T细胞的构建及其对多发性骨髓瘤细胞的体 外杀伤效果[J].中国肿瘤生物治疗杂志, 2024, 31(12): 1186-1193.
Jin C, Deng J, Jiang Y, Zhu J, Kang L, Li S. BCMA CAR-T therapy as salvage therapy in patients with plasmablastic myeloma. Hematology. 2025 Dec;30(1):2481555. doi: 10.1080/16078454.2025.2481555. Epub 2025 Mar 27. PMID: 40146876.
https://www.ncbi.nlm.nih.gov/books/NBK584177/
Banerjee R, Richards A, Midha S, Afrough A, Anwer F, Atanackovic D, Atrash S, Bachanova V, Beitinjaneh AM, Bhurtel E, Castaneda Puglianini O, Chhabra S, Cicero KI, Davis JA, Dhakal B, Dima D, Ferreri CJ, Forsberg PA, Freeman CL, Herr MM, Jain T, Janakiram M, Khouri J, Kocoglu MH, Kumar A, Liu Y, Locke F, McGuirk JP, Mikkilineni L, Nadeem O, Parrondo RD, Pasvolsky O, Peres LC, Purvey S, Raza S, Reshef R, Richard S, Rossi AC, Sborov DW, Shune L, Wagner CB, Zanwar SS, Sidana S, Patel KK, Hansen DK, Kumar SK, Lin Y, Martin TG, Voorhees PM, Anderson LD Jr, Cowan AJ, Kaur G. Universal driving restrictions beyond 4 weeks appear unnecessary following CAR-T therapy in multiple myeloma. Blood Adv. 2025 May 13;9(9):2336-2340. doi: 10.1182/bloodadvances.2025016131. PMID: 40088469; PMCID: PMC12127628.














