Overview
Chimeric Antigen Receptor T-Cell Therapy (CAR-T) is a cutting-edge form of cellular immunotherapy. This innovative treatment re-engineers a patient’s own T lymphocytes through genetic modification, enabling them to precisely recognize and target tumor cells for elimination.
The advent of CAR-T therapy has brought new hope for patients with hematologic malignancies and is showing promising potential in treating autoimmune diseases and solid tumors—opening new therapeutic avenues for individuals who previously lacked effective treatment options.
Applications of CAR-T Therapy
CAR-T therapy has demonstrated remarkable efficacy in multiple hematologic malignancies, including acute lymphoblastic leukemia (ALL), lymphoma, and multiple myeloma. Meanwhile, its applications are being actively explored in autoimmune diseases such as systemic lupus erythematosus and in solid tumors such as gastrointestinal cancers, offering new possibilities for broader patient populations.
GoBroad Healthcare Group has established a comprehensive CAR-T clinical research and treatment system, covering every stage from patient screening, cell manufacturing, and treatment administration to long-term follow-up. We not only provide routine CAR-T treatments for various hematologic malignancies but also actively advance pioneering clinical studies in autoimmune diseases and solid tumors—committed to extending the benefits of cellular immunotherapy to a wider range of patients.
Why Choose GoBroad?
GoBroad Healthcare Group was among the first in China to systematically develop and implement CAR-T cell therapy, continuously promoting the standardization and clinical innovation of this technology. Having performed several thousand CAR-T treatments, our team has accumulated extensive experience and achieved clinically validated efficacy and safety across diverse disease types.
Comprehensive Target Portfolio
GoBroad has built a mature CAR-T technology platform with an extensive range of therapeutic targets, enabling precision treatment from hematologic malignancies to solid tumors.
Targets for Hematologic Tumors:
- Single-target CAR-T: CD19, CD20, CD22, CD7, CD5, BCMA, GPRC5D, CLL1
- Dual-target CAR-T: Sequential CD19-22 CAR-T, Sequential CD19-20 CAR-T
Targets for Solid Tumors:
- Claudin18.2 CAR-T and others
Innovative Combination Strategies
GoBroad actively integrates CAR-T therapy with various conventional and emerging treatments—including chemotherapy, targeted therapy, and hematopoietic stem cell transplantation—to improve overall remission rates and long-term survival in relapsed or refractory patients.
Comprehensive Treatment and Care Management
Our integrated care model encompasses assessment, implementation, monitoring, and follow-up throughout the entire CAR-T treatment journey. From patient enrollment and cell preparation to therapy execution and rehabilitation, GoBroad provides personalized, systematized care supported by multidisciplinary collaboration to ensure safety, efficacy, and quality of long-term follow-up.
Multidisciplinary Collaboration (MDT)
GoBroad practices a patient-centered, multidisciplinary team approach, uniting hematology, integrated diagnostics, infectious disease imaging, respiratory medicine, and other specialties. This cross-disciplinary collaboration enables precise, systematic, and comprehensive diagnosis and treatment, forming the core strength of our cli
Our Achievements
For Relapsed/Refractory Leukemia
- CD19 CAR-T therapy for R/R B-ALL: Complete remission (CR) rate over 90%
- Sequential CD19 and CD22 CAR-T post-transplant relapse (B-ALL): 12-month and 18-month overall survival (OS) rates of 88.5% and 67.5%, respectively
- CD22 CAR-T after CD19 CAR-T failure: CR rate 71%, MRD negativity near 100%, outperforming international benchmarks
- Sequential CD19 and CD22 CAR-T in pediatric R/R B-ALL (Phase II): 18-month event-free survival (EFS) 79%, disease-free survival (DFS) 80%, OS 96%
- CD19 CAR-T for relapsed/refractory non-B-cell acute leukemia (AML, T-ALL): CR rate 62.5%
- Donor-derived CD7 CAR-T for R/R T-ALL: CR rate 90%, with 27-month median follow-up showing ORR 95% and CR 85%
- Donor-derived CD5 CAR-T for R/R T-ALL: 100% achieved CR or CRi by day 30 post-infusion
- Autologous unselected CD7 CAR-T for R/R T-ALL: 96% response rate, 85% achieving CR or CRi by day 30
For Relapsed/Refractory Lymphoma and Multiple Myeloma
- First global report: Triple-target CD19/CD20/CD22 CAR-T therapy in pediatric R/R Burkitt lymphoma achieved a 100% response rate and 86.9% CR at 18 months
- Autologous HSCT combined with CAR-T for R/R CNS B-cell lymphoma: ORR 74%, 1-year ORR 85.1%, 1-year PFS 44.5%, with controllable ICANS
- Donor CAR-T as preconditioning for allo-HSCT in R/R B-cell lymphoma: Median follow-up 249 days; CR 75%, 6-month OS and PFS 75% and 62.5%, respectively
The efficacy data are derived from published studies and GoBroad Healthcare Group’s long-term follow-up results. For medical reference only. Individual outcomes may vary depending on disease type and treatment plan. Please consult with a qualified physician for a personalized treatment strategy.
Related Reading
Relapsed/refractory central nervous system lymphoma (R/R CNSL) is associated with poor survival outcomes, which vary significantly depending on multiple factors. Among them, TP53 mutation is considered one of the key determinants of treatment response and has long posed a clinical challenge. In recent years, chimeric antigen receptor T-cell (CAR-T) therapy has demonstrated promising efficacy and safety in R/R CNSL. However, limited data are available regarding whether TP53 mutations influence the effectiveness of CAR-T therapy, warranting further investigation.
Recently, the lymphoma and myeloma team at GoBroad Healthcare Group, led by Professor Ke Xiaoyan and Professor Hu Kai, published a study entitled “CAR-T cell therapy in TP53-mutated CNS lymphoma: overcoming a high-risk genetic barrier” in Frontiers in Medicine. The first author of the paper is Li Danyang. This study provides new insights into CAR-T therapy for TP53-mutated R/R CNSL.
Professor Hu Kai explained that the treatment of R/R CNSL remains challenging. Patients who are sensitive to chemotherapy may achieve remission and benefit from autologous hematopoietic stem cell transplantation. However, in real-world practice, many patients relapse and develop chemotherapy resistance, particularly those harboring TP53 mutations, resulting in poor prognosis. The team retrospectively analyzed 61 patients with R/R CNSL, 43 of whom received CAR-T therapy. Overall survival (OS) and progression-free survival (PFS) were compared between patients with TP53 mutations (TP53+) and those with wild-type TP53 (TP53−), and prognostic factors were evaluated.
Key Findings
1. Comparable efficacy of CAR-T therapy in TP53+ and TP53− R/R CNSL patients In the TP53+ group, the overall response rate (ORR) and complete response rate (CRR) were both 64.5%. In the TP53− group, the ORR and CRR were 73.3% and 69.2%, respectively, indicating similar response rates between the two groups. The median PFS in the TP53+ group was 12.77 months (95% CI: 6.33–∞), compared with 22.4 months (95% CI: 6.13–∞) in the TP53− group. Although numerically shorter, the difference was not statistically significant. These results suggest that CAR-T therapy is effective in R/R CNSL patients with TP53 mutations.
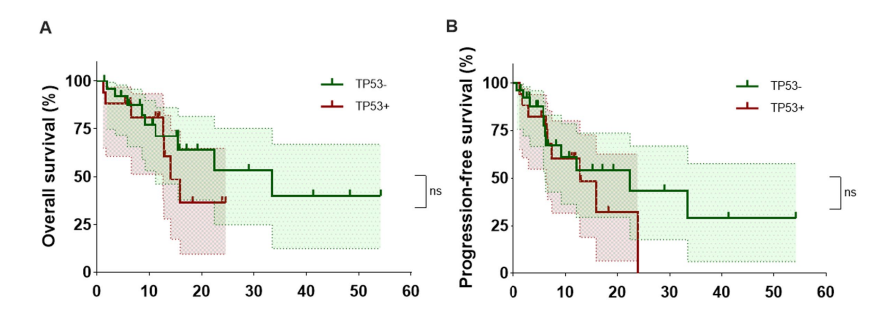
2. Potentially greater benefit in non-GCB TP53+ R/R CNSL patients Subgroup analysis demonstrated that the cell-of-origin (COO) classification significantly influenced long-term survival. In the TP53+ cohort, patients with the non-germinal center B-cell-like (non-GCB) subtype had significantly longer OS than those with the GCB subtype (P = 0.003).
The preliminary findings from this center indicate that CAR-T therapy represents an effective treatment option for TP53-mutated R/R CNSL patients and may offer particular benefit for those with the non-GCB subtype. These data provide valuable clinical reference for treatment selection.
Conclusion and Future Perspectives
Professor Ke Xiaoyan noted that the lymphoma and myeloma department at GoBroad Healthcare Group has accumulated extensive experience in CAR-T therapy. As the number of real-world CAR-T cases increases, clinical management has become more complex. Clinical experience suggests that patients with TP53 mutations often exhibit high resistance to chemotherapy, whereas CAR-T therapy may provide meaningful benefit. Optimizing the timing of CAR-T therapy and implementing individualized, refined, and comprehensive management strategies remain key objectives in hematologic oncology.
Future approaches may integrate clinical, pathological, molecular, and imaging diagnostics to identify R/R CNSL patients who are resistant to conventional therapies but likely to benefit from CAR-T treatment. In addition, monitoring circulating tumor DNA (ctDNA) may enable earlier detection of relapse and timely intervention, ultimately prolonging survival.


Marina is a 61-year-old woman from Russia. After being diagnosed with acute T-lymphoblastic leukemia (T-ALL), she began a long and challenging course of treatment.
When Challenges Returned After Transplant
In April 2025, Marina underwent an allogeneic hematopoietic stem cell transplant in her home country. For her and her family, it was an important step forward and carried hope for lasting remission.
Three months later, follow-up testing revealed measurable residual disease (MRD) in her bone marrow, with a small but concerning level of malignant cells (0.014%). Immunotherapy was attempted locally, but the disease continued to show signs of progression. At the same time, Marina developed mild skin graft-versus-host disease.
For an older patient facing immune-related relapse after transplantation, treatment options are limited and the overall situation becomes significantly more complex.
Re-evaluating Treatment in China
After careful consideration, Marina traveled to China for further evaluation. A multidisciplinary team led by Dr. Zhao Defeng at GoBroad Healthcare Group performed a comprehensive reassessment of her disease. The results showed that her leukemia cells predominantly expressed the CD7 antigen.
Following detailed discussions with Marina, the team first initiated immuno-targeted therapy to stabilize her condition. Based on her disease profile and treatment history, a decision was then made to proceed with CD7 CAR-T cell therapy.
The GoBroad team has extensive experience treating T-cell malignancies with CAR-T cell therapy, including complex cases involving older patients and post-transplant relapse, allowing for highly individualized risk assessment and clinical management.
Navigating Treatment and Side Effects
After CAR-T cell infusion, Marina developed a high fever as the infused cells began to expand in her body. Her main side effect was cytokine release syndrome (CRS), primarily presenting as fever, along with a temporary drop in oxygen levels.
Given her underlying heart and lung conditions, the medical team closely monitored her and intervened promptly when needed. With careful and standardized management, Marina successfully passed through the CRS phase without serious complications.
A Meaningful Milestone
On day 13 after CAR-T infusion, a bone marrow examination showed that MRD was no longer detectable, indicating a deep remission.
At the same time, next-generation sequencing (NGS) covering more than 300 genes identified eight previously undetected genetic mutations. These findings provided valuable guidance for future treatment planning, including the possibility of targeted therapies.
Looking Ahead
With a positive response achieved, Marina plans to return home to prepare for further stem cell transplantation. Based on her individual risk profile, the medical team recommended early post-transplant maintenance therapy with targeted agents to help reduce relapse risk and support long-term disease control.
A Journey Still Unfolding
Marina’s treatment journey has been anything but easy. She has faced transplantation, relapse, and difficult decisions involving high-risk therapies. Through careful evaluation, thoughtful planning, and close collaboration between patient and medical team, she has now reached an important turning point.
Her story is still unfolding—guided by caution, experience, and a shared commitment to achieving the best possible long-term outcome.


At GoBroad Shanghai Liquan Hospital, Prof. Su Li shared his valuable clinical experience, research achievements, and forward-looking perspectives in the treatment of multiple myeloma .
Q1. While BCMA-targeted CAR-T therapy has achieved breakthroughs in multiple myeloma, plasmablastic myeloma (PBM), as an aggressive subtype, has long lacked effective treatment options. What is the current status of diagnosis and treatment for PBM patients?
Prof. Su Li: Plasmablastic myeloma is a special subtype of myeloma with unique clinical characteristics. In clinical practice, a small proportion of patients can be diagnosed at disease onset, but the majority are only identified at the relapsed/refractory stage. The overall incidence remains relatively low, though it varies among centers. Our research has shown an incidence of approximately 3%–18%.
In terms of treatment, current outcomes for PBM remain unsatisfactory. Neither traditional chemotherapy nor novel therapeutic strategies have significantly improved remission or long-term survival. Existing clinical data suggest a median overall survival of about 10 months (range 6–12 months) for most patients, underscoring the urgent need for more effective approaches to improve prognosis and break through current clinical limitations.
Q2. How do the outcomes of BCMA CAR-T monotherapy compare with BCMA CAR-T combined with transplantation in PBM patients? What factors are usually considered when formulating a treatment strategy?
Prof. Su Li: The application and progress of CAR-T therapy in PBM must be understood within its biological context and clinical practice. Our earlier studies found that BCMA expression rises significantly in refractory/relapsed myeloma, especially with extramedullary involvement, providing a clear biological basis for BCMA-targeted CAR-T therapy.
Studies comparing single-target versus multi-target CAR-T strategies have shown that single-target approaches often result in early extramedullary relapse. In contrast, dual-target regimens, such as BCMA/CD19 or BCMA/GPRC5d, achieve higher MRD-negativity rates, deeper responses, and more durable remissions compared to BCMA monotherapy.
In clinical practice, CAR-T selection requires a comprehensive assessment of several factors. Pathological testing (immunohistochemistry, bone marrow flow cytometry, etc.) must first confirm antigen expression. For patients with BCMA-only expression, a BCMA/CD19 dual-target CAR-T is preferable. For those co-expressing BCMA and GPRC5d, a BCMA/GPRC5d dual-target approach is recommended. Dual-target regimens are especially advantageous in extramedullary disease, whereas single-target therapy may still be effective in purely intramedullary disease.
Another critical factor is prior CAR-T exposure. With growing adoption, some patients relapse after initial CAR-T therapy, often due to antigen loss or residual CAR-T immunogenicity, leading to diminished efficacy. For relapsed patients, switching to a CAR-T with a different construct is essential to avoid immune interference. Clinical practice has confirmed that such heterologous construct selection yields significant efficacy in short-term relapsed or expansion-failure patients, demonstrating its value in real-world treatment.
Q3. What challenges remain in applying CAR-T therapy more broadly in multiple myeloma, and how might they be overcome to benefit more patients?
Prof. Su Li: As a rare disease, PBM has a low incidence, and large-scale international studies are scarce. CAR-T therapy still faces several challenges:
- Accessibility: High costs and frequent out-of-pocket payment limit widespread adoption. Future innovations in technology or payment models could lower costs, enabling CAR-T to be used earlier rather than only at terminal stages. Our research shows that most patients receive CAR-T late in the disease course, where efficacy is more limited.
- Timing: Treatment line strongly affects outcomes. Later-line patients have weaker immune function, and their harvested T cells are often senescent or functionally impaired, reducing CAR-T manufacturing quality and in vivo expansion. Early T-cell collection, when cells are healthier, could improve outcomes.
- Construct design: Efficacy varies among patients even with the same CAR-T product. Beyond T-cell quality, antigen-binding affinity between CAR-T and tumor cells plays a role. Continued optimization of CAR design to enhance antigen recognition and binding is a key research focus.
- Combination strategies: Sequential or combinatorial approaches are under active investigation, such as CAR-T followed by autologous stem cell transplantation, or maintenance with immunomodulators or PD-1 inhibitors. These strategies may prolong remission and improve survival, and related studies are ongoing.


Decoding the Blueprint of Life: CAR-T as a Precision Navigation System Starting from Hematologic Malignancies
In the vast landscape of cancer immunotherapy, CAR-T cell therapy represents a transformative innovation. According to Dr. Changsong Qi of Peking University Cancer Hospital and Beijing GoBroad Hospital, its remarkable efficacy in hematologic malignancies stems from highly specific tumor-associated antigens such as CD19 (in acute lymphoblastic leukemia) and BCMA (in multiple myeloma), which serve as precise "navigation systems" for T cells.
"Essentially, the CAR construct reprograms T cells, enabling them to identify and eliminate tumor cells," explained Professor Qi. Once reinfused, these engineered T cells are able to precisely target and destroy tumor cells expressing these markers, achieving high response rates and sustained immune surveillance — a principle now validated by multiple commercial products and national regulatory frameworks.
The Challenge of Solid Tumors: Redefining the Role of CAR-T Therapy
Compared with hematologic cancers, the treatment of solid tumors is significantly more challenging, primarily due to:
- The lack of ideal, broadly expressed tumor-specific targets
- A highly immunosuppressive tumor microenvironment
- Tumor heterogeneity and antigen escape
“We initially explored targets like EGFR, but early studies showed an objective response rate of less than 10%, coupled with severe toxicity,” Professor Qi acknowledged. However, breakthroughs have since emerged with novel targets in solid tumors — including Claudin18.2 (gastric cancer), GPC3 (hepatocellular carcinoma), and GUCY2C (colorectal cancer).
Claudin18.2 has proven to be one of the most promising targets in gastric cancer, with expression in nearly 60% of moderate-to-high Claudin18.2-positive cases. CT041, a CAR-T product developed against this target, has entered Phase II clinical trials. Among over 200 patients treated, the objective response rate has consistently ranged between 40–50% — more than tenfold higher than the 2–5% seen with third-line standard treatments.
In liver cancer, GPC3 is expressed in approximately 70% of hepatocellular carcinomas and is nearly absent in normal tissues, making it an ideal “window target” for CAR-T therapy. New generations of structurally optimized GPC3-CAR-T products have achieved objective response rates exceeding 70% in mid-to-high dose cohorts, significantly prolonging median survival.
For colorectal cancer, GUCY2C (also known as GCC) shows stable expression in 60–70% of patients across both primary and metastatic sites. Professor Qi's team is collaborating with industry partners to advance CAR-T products targeting GUCY2C, with early studies showing objective response rates of approximately 40%.
Innovative Delivery Routes: Paving the Way for CAR-T Cells to Reach Solid Tumors
A key determinant of CAR-T efficacy lies in the ability to physically deliver cells to tumor sites. “Traditional intravenous infusion often proves inadequate for certain locations,” Professor Qi emphasized. “Targeted delivery strategies are essential.”
Several innovative administration routes are currently being explored in clinical settings:
- Intraventricular injection: For intracranial tumors like neuroblastoma, CAR-T cells are infused directly into the brain ventricles to bypass the blood-brain barrier, enhance local effects, and minimize systemic toxicity.
- Hepatic arterial infusion: Especially for liver cancers expressing GPC3, CAR-T cells are delivered through the hepatic artery to achieve high local concentrations.
- Peritoneal/pleural infusion: Applicable to peritoneal metastases or thoracic tumors to maximize local efficacy.
In combination with debulking surgery, these approaches reduce tumor burden and improve the tumor microenvironment, ultimately enhancing CAR-T cell persistence and function.
Clinical Cases: Witnessing the Power of a “Living Drug”
Dr. Qi shared several compelling clinical cases that vividly illustrate the life-altering potential of CAR-T therapy as a “living drug”:
- A gastric cancer patient with anastomotic recurrence who was unable to eat regained the ability to consume porridge within just over a week following Claudin18.2-CAR-T infusion. One month later, rapid tumor regression even triggered bleeding — a dramatic confirmation of CAR-T's potent cytotoxicity.
- Another patient, previously fully reliant on parenteral nutrition due to complete obstruction, resumed oral intake just seven days after treatment and went on to enjoy over two years of high-quality life.
- A woman in her 30s, suffering from peritoneal metastases, renal failure, and carrying four drainage tubes, had all tubes removed within six months post-treatment and returned to work.
These examples demonstrate that CAR-T therapy is evolving from an experimental intervention into a modality capable of reconstructing lives.
Common Questions Answered: Helping More Patients Take the First Step
At the end of the interview, Professor Qi addressed several of the most pressing questions raised by patients:
On eligibility for clinical trials:
“Not all patients can enter clinical studies immediately,” he emphasized. “Target antigen testing is essential. Only when expression is confirmed can tumor burden and metastatic status be evaluated for trial inclusion.”
On 'pseudo-progression' post-treatment:
Some patients may initially show tumor enlargement on imaging. This is often due to CAR-T cell infiltration and inflammation rather than true disease progression. Imaging density and clinical symptoms are key to distinguishing between the two.
On timing and treatment duration:
“Unlike chemotherapy, CAR-T therapy involves a single infusion,”Professor Qi explained. “If the patient is stable, we recommend allowing time for the cells to act, rather than rushing into further treatment decisions.”
Closing Words: From Breakthrough to Cure, the Future of CAR-T Therapy Is Worth Pursuing
Dr. Qi concluded with an encouraging message:“Today, we are no longer content with simply prolonging survival — we are striving for true functional cures. CAR-T therapy is opening up new possibilities in the treatment of solid tumors. As long as we keep exploring and hold on to hope, the dawn of better outcomes will surely come.”


Jing Pan, Yue Tan, Lingling Shan, Samuel Seery, Biping Deng, Zhuojun Ling, Jinlong Xu, Jiajia Duan, Zelin Wang, Kai Wang, Xinjian Yu, Qinlong Zheng, Xiuwen Xu, Guang Hu, Taochao Tan, Ying Yuan, Zhenglong Tian, Fangrong Yan, Yajing Han, Jiecheng Zhang & Xiaoming Feng
Abstract
Refractory or relapsed T cell acute lymphoblastic leukemia (r/r T-ALL) patients have poor prognoses, due to the lack of effective salvage therapies. Recently, CD7-targeting chimeric antigen receptor (CAR)-T therapies show efficacy in patients with r/r T-ALL, but relapse with CD7 loss is common. This study evaluates a CD5-gene-edited CAR-T cell therapy targeting CD5 in 19 r/r T-ALL patients, most of whom had previously failed CD7 CAR-T interventions. CAR-T products were derived from previous transplant donors (Cohort A) or newly matched donors (Cohort B). Primary endpoints were dose-limiting toxicity at 21 days and adverse events within 30 days. Secondary endpoints were responses, pharmacokinetics and severe adverse events after 30 days. A total of 16 received infusions, 10 at target dose of 1 × 106 kg−1. All encountered grade 3–4 cytopenias and one had a grade 3 infection within 30 days. All patients (100%) achieved complete remission or complete remission with incomplete blood count recovery by day 30. At a median follow-up of 14.3 months, four received transplantation; three were in remission and one died of infection. Of 12 untransplanted patients, 2 were in remission, 3 relapsed, 5 died of infection and 2 of thrombotic microangiopathy. CAR-T cells persisted and cleared CD5+ T cells. CD5− T cells, mostly CD5-gene-edited, increased but remained below normal levels. These results suggest this CD5-specific CAR-T intervention has a high remission rate for T-ALL patients. Evidence also suggests the risk of late-onset severe infection may be mitigated with consolidative transplantation. This study provides insights that could help to optimize this promising intervention.
Refer to the original: https://doi.org/10.1038/s41591-024-03282-2


















